Custom Property Types
It seems you can never have enough Metadata. Last month I wrote about Custom Metadata for Projects and Drafts in the TrialGrid system and this week we expanded this capability further. You can now define Custom Properties for Forms, Fields, Projects and Drafts in the TrialGrid system and for each of those Properties define its display order, data type (text, boolean or choice) and list of allowed values.
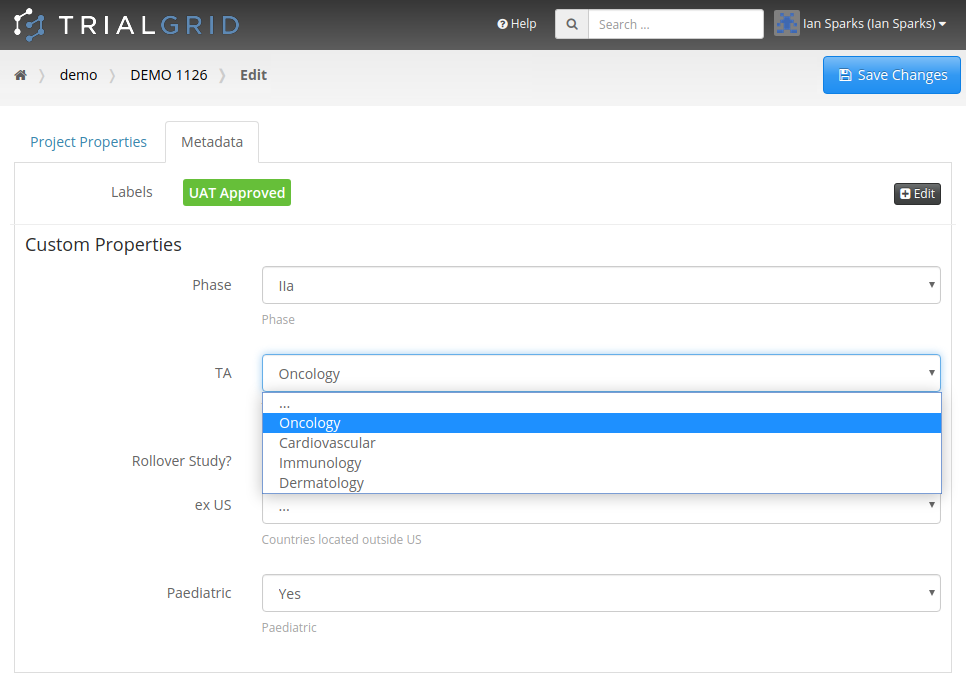
Our industry is increasingly turning to Metadata Repositories (MDRs) to manage study design elements. Part of the promise of these systems is to capture not just the "What" of Forms, Fields, Data Dictionaries and other design elements but also the "Why" and the "How" : Why should this Field appear in this Form in this type of study? and How should the Form be modified if this is a Phase II rather than a Phase III study?
For all it's strengths, Medidata Rave Architect does not have any capability to import custom metadata. An MDR can output an Architect Loader Spreadsheet which defines the structure of the study but there's no way to include any of the data that led to that particular configuration. Examples include:
- Is this a follow-on study?
- What is the age group of the study participants?
- Is the study blinded, unblinded?
- What is the therapeutic area for this study?
- Will the study run in the USA / Europe / China / Japan?
- What Medical Coding system will be used for the study?
- Does the study use Rave Safety Gateway?
- Is there an ePRO component to the study?
Having this information associated as data to the study build is very useful to study builders in the short term but we have ambitions to use this metadata to automatically configure and validate study build. Watch this space!