Next week we're attending the Medidata Next conference in New York and we're
looking forward to meeting up with friends old and new, customers and past colleagues. In the run up to the conference
we're working on major new features but that doesn't mean we're neglecting existing functionality.
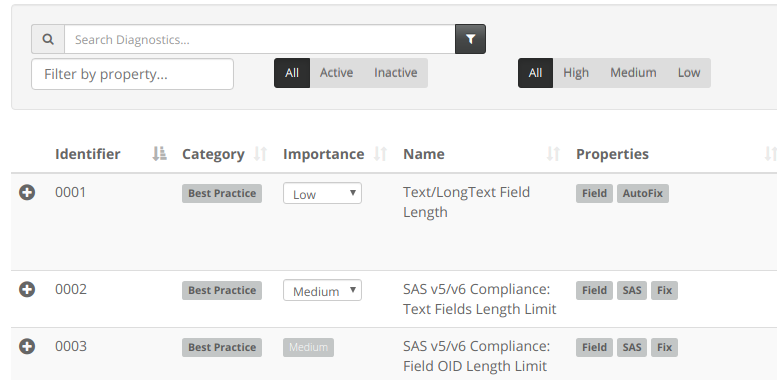
This week we've been spending more quality time with Diagnostics.
New Diagnostics
Our policy has always been that if a Diagnostic is suggested to us by a customer and we think it makes sense for the
community we'll add it to our catalog at no cost. A customer contacted us to suggest a diagnostic for Static Values in
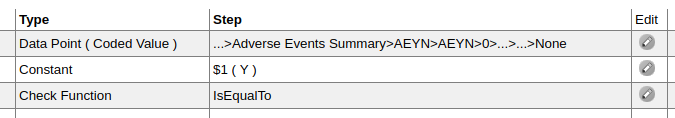
Edit Check steps. As it stands, Rave believes you know what you are doing and will allow you to do this:

Here I'm defining a Static Value as "NONE" with a format of $3. That's 3 characters which means Rave will
interpret this value as "NON". Something is wrong here. Either the value should be "NON" or the format should be $4
so that Rave will see "NONE". Diagnostic 86 identifies this situation and it happens more often than you would expect.
Meanwhile another customer was also in touch to suggest a new diagnostic for Form level Signature Is Required. The issue is that
if a form is set as Signature Required = No (unchecked) but there are Fields which Participate in Signature on the Form
then Fields may not be able to be locked because they have not been signed. Diagnostic 85 identifies this situation.
Conversely, you can have a Form which is Signature Required = Yes (checked) but there are no Fields on the Form which
actually participate in Signature. Probably you don't need to sign a page where there are no Fields which can be
signed so Diagnostic 87 identifies this state of affairs.
Previously, this customer had suggested diagnostics for Data Entry and Non-Data Entry Fields. For example,
a Field which cannot be data entered by a user should not have an OpenQuery action on it because the user would not
be able to make changes to the Field to action that query. Similarly, a field where you expect data entry by
Investigator users should be visible. You might also want to make sure these Fields are verified and require data entry.
In all, these Data-Entry and Non-Data Entry diagnostics bring us to 94 diagnostics in total.
Updates to existing Diagnostics
Diagnostic 71 is designed to find Required Fields which are view/entry restricted from Site Users
"Site Users" means Investigators, Study Coordinators and other roles who are expected to provide the values for these
required fields. To assist with this diagnostic TrialGrid has a flag in its URL configuration for EDC roles which
determines if an EDC role represents a "Site User". This is useful but not flexible enough for some users so this
week we added a new feature and updated Diagnostic 71 with a selector for role names and exclusions.

Entering or selecting Role names in this setting will override the "Site User" flag in the configuration for this
diagnostic. Note the color coding of Role names. If a name appears in the configuration it appears in Blue, in Orange
if it is not in the configuration. This can be important when you are copying diagnostics settings between Projects
in different URLs.
New Diagnostic Features
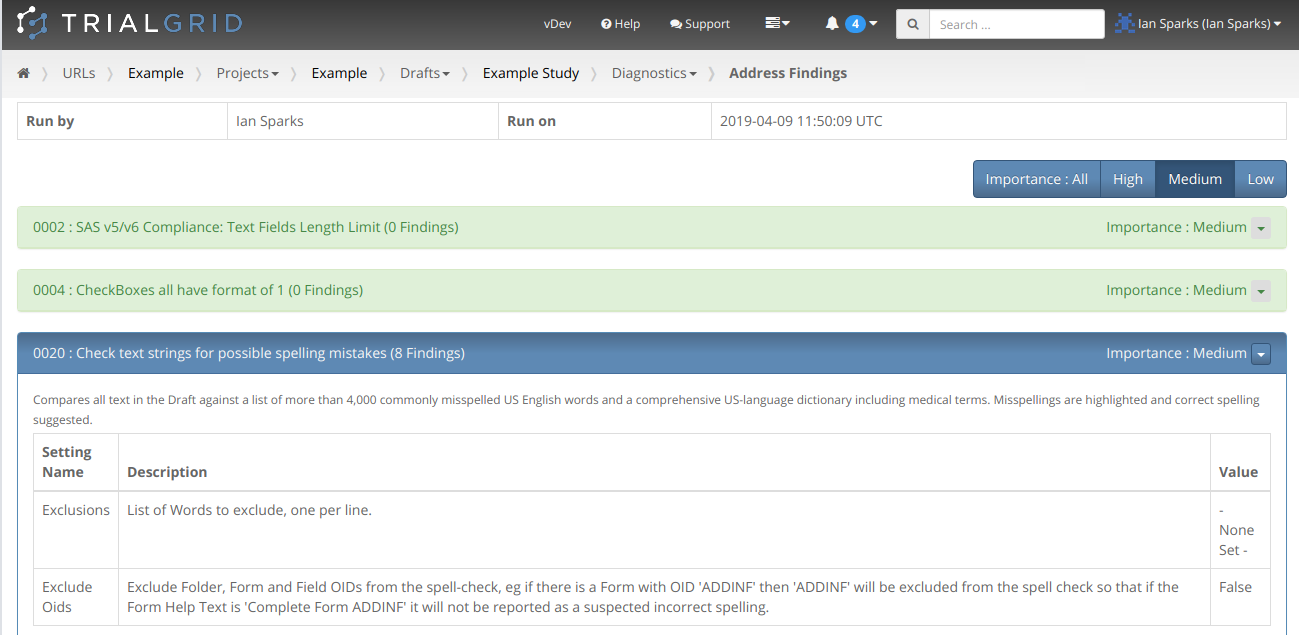
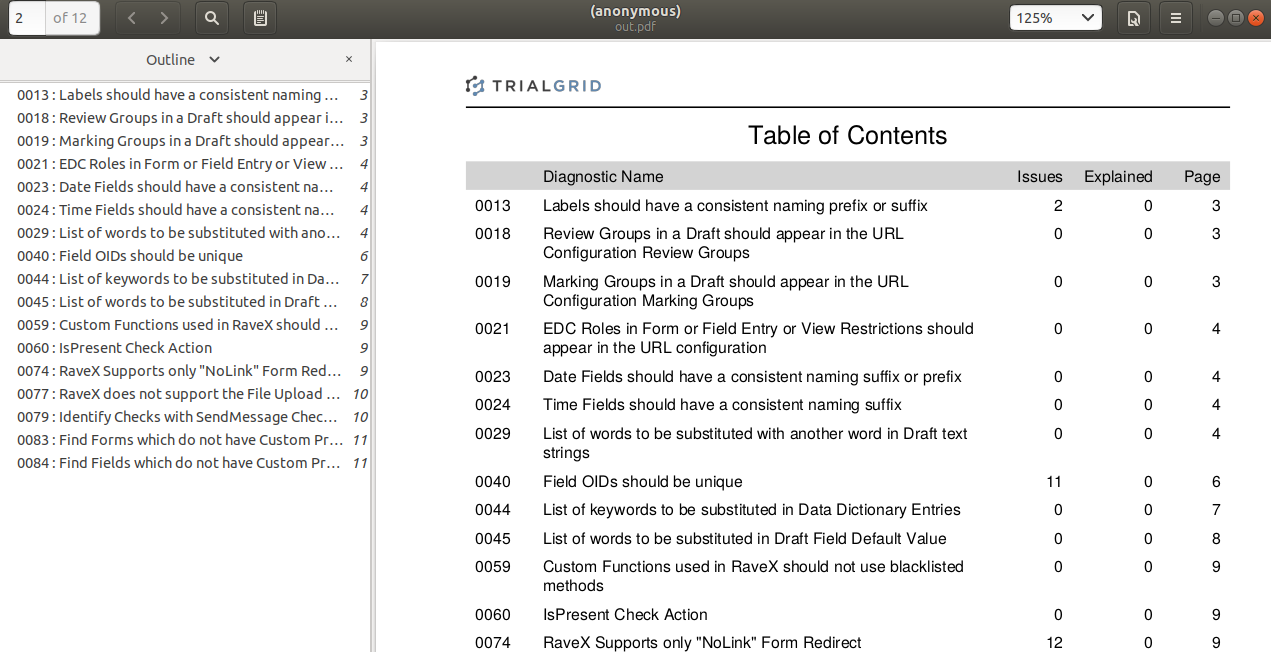
We recently heard from a group evaluating TrialGrid that a table of contents and bookmarking in the PDF diagnostic
report would be a useful feature, especially where there are a lot of findings. We agreed and from now on
all PDF diagnostic reports will include a Table of Contents page with hyperlinks to the results of each diagnostic
from the PDF outline view and from the page numbers in the Table.
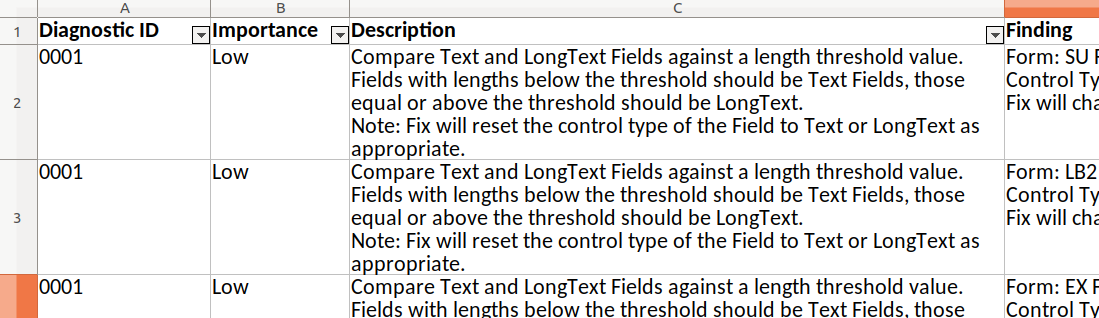
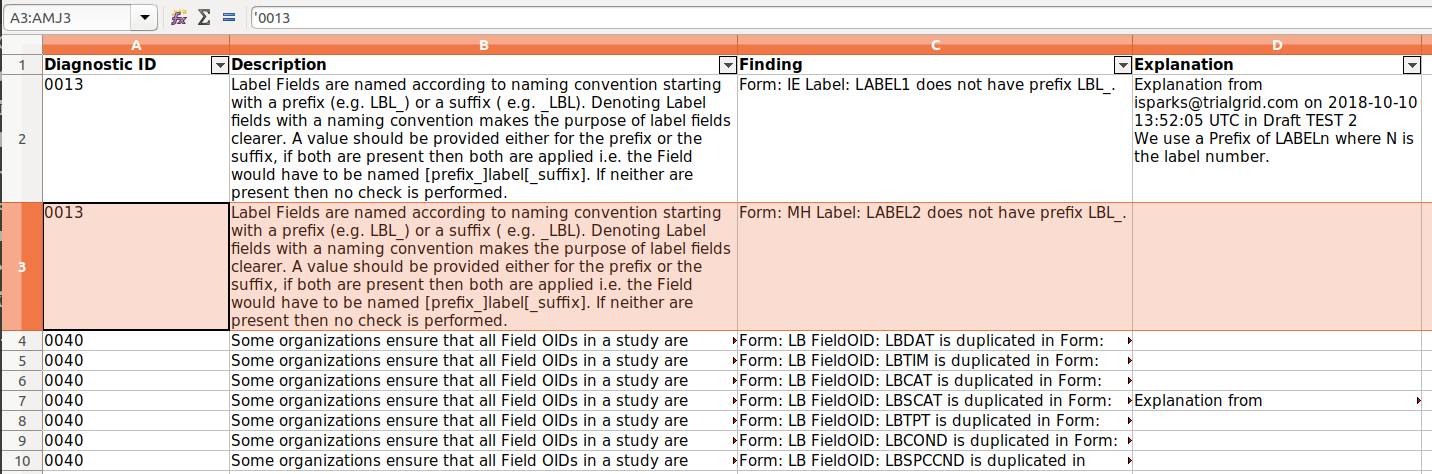
But sometimes reviewing diagnostic results would be easier if you had them in an easily sortable and filterable format
like a spreadsheet. To support that we added a new report to the diagnostic process - an Excel spreadsheet of the
results:
Summary
Last week we added the Search Feature to quickly find objects across all your study designs. This week it has been
new and updated diagnostics and new diagnostic features. The TrialGrid product continues to improve, in large part
driven by customer feedback so a big thank you to everyone who has contributed ideas.
Next week we'll be in New York for the Medidata Next conference and we look forward to seeing you there!